Zeewier en bierbrouw-afval blijken volwaardige vervangers van schadelijke pesticiden
Om gewassen te beschermen tegen ziekteverwekkers worden deze in de moderne landbouw vaak bespoten met verschillende pesticiden. Dit zijn vaak giftige, niet biologisch afbreekbare bestanddelen die, wanneer zij via neerslag en wind in het milieu terechtkomen, biodiversiteit verminderen. Pesticideresiduen vormen ook een gezondheidsrisico wanneer zij in hoge concentraties in voedsel aanwezig zijn. Om afhankelijkheid te verminderen kunnen biostimulanten een vervanger vormen, dit zijn biologische stoffen die een versterkend effect kunnen hebben op de behandelde plant, waardoor de weerstand tegen pathogenen mogelijks verhoogt. Deze stoffen zijn biologisch afbreekbaar en onschadelijk voor het milieu, wat in overeenstemming is met de "Farm to Fork" EU-strategie om voedselsystemen eerlijk, gezond en milieuvriendelijk te maken.
Welke stoffen werden getest?
In dit artikel werden de modelplant Arabidopsis thaliana en de biotrofe plantpathogeen Hyaloperonospora arabidopsidis gebruikt om meeldauwinfecties te simuleren. Zes biostimulanten werden beoordeeld op hun effect op de proliferatie van het pathogeen, namelijk humuszuur (HA1), fulvinezuur (HA2), soja-extract (PH1), moutextract (PH2) en Ecklonia maxima zeewierextract van twee leveranciers (SW1) en (SW2). De doeltreffendheid van deze biostimulanten werd geëvalueerd in vier verschillende experimenten en werd vergeleken met een waterbehandeling als negatieve controle en met een difenoconazol houdend fungicide als positieve controle.
Hoe werden de stoffen getest?
Het effect van de biostimulanten op geïnfecteerde A. thaliana na behandeling werd gekwantificeerd door het tellen van H. arabidopsidis-sporen via microscopie en het kwantificeren van de relatieve pathogene biomassa via quantitative polymerase chain reaction (qPCR). Verder werd een pigmentextractie uitgevoerd op geïnfecteerde planten om chlorofyl- en carotenoïdengehalten te bepalen via spectrometrie. Zo werd het absolute chlorofylgehalte gebruikt als maat voor de groei en biomassa van de plant, het relatieve chlorofylgehalte als maat voor fotosynthesecapaciteit, en het relatieve carotenoïdengehalte als maat voor immuunrespons. Een laatste experiment werd uitgevoerd met de twee best presterende biostimulanten uit voorgaande experimenten, waarbij groeicurven en kinetische parameters van zowel sporulatie als relatieve pathogene biomassa werden opgesteld.
Zeewierextract en moutextract blijken mogelijke volwaardige vervangers
Uit deze experimenten bleek dat moutextract (PH2) en Ecklonia maxima zeewierextract van Kelpak® (SW2) de beste prestaties leverden. Beide behandelingen lieten consistent een significante vermindering zien van H. arabidopsidis-sporen en biomassa, vergelijkbaar met het fungicide. Beide behandelingen lieten ook verbeteringen zien in de groei en fotosynthesecapaciteit van de planten, vergelijkbaar met die van niet-geïnfecteerde planten, en een verhoogde immuunrespons vergeleken met de waterbehandeling. Bovendien veroorzaakte PH2, net als het fungicide, een vertraagde en verminderde sporulatiepiek en een vervroegde verminderde H. arabidopsidis biomassapiek.
Maar…
Hoewel deze biostimulanten veelbelovende resultaten opleverden, is er nog ruimte om concentraties en toedieningsmethoden te optimaliseren om de doeltreffendheid te verhogen. Ook dienen er veldproeven worden uitgevoerd om een indicatie te krijgen van de doeltreffendheid op productieschaal. Indien deze biostimulanten met succes worden geïntegreerd, kunnen zij leiden tot een wereldwijde verschuiving naar duurzamere landbouwpraktijken, met positieve gevolgen voor het milieu, biodiversiteit en voedselkwaliteit.
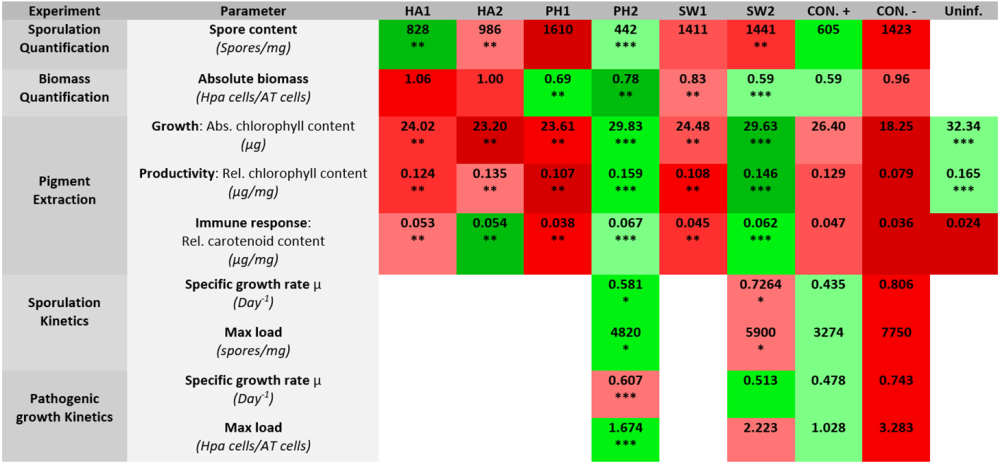
Tabel: Overzicht van alle experimentele resultaten. In de eerste kolom staan alle experimenten. In de tweede kolom staan alle experimentele parameters. Deze worden gevolgd door de respectieve gemiddelde resultaten van alle behandelingen voor die parameter, waarvan de 3 best presterende behandelingen worden weergegeven in groene tinten en de slechtst presterende behandelingen in rode tinten. Hoe lichter de tint, hoe beter de prestatie van de biostimulant voor de betreffende parameter. Behandelingen met een significant verschil ten opzichte van de negatieve controle (CON.-) worden aangegeven met (*), behandelingen die niet significant verschillen van de positieve controle (CON.+) worden aangegeven met (**), en behandelingen die aan beide criteria voldoen worden aangegeven met (***).
Bibliografie
A. C., Schürmann, P., & Buchanan, B. B. (1976). Effect of powdery mildew infection on photosynthesis by leaves and chloroplasts of sugar beets. Plant Physiology, 57(4), 486-489.
Al-Huqail, A., El-Dakak, R. M., Sanad, M. N., Badr, R. H., Ibrahim, M. M., Soliman, D., & Khan, F. (2020). Effects of climate temperature and water stress on plant growth and accumulation of antioxidant compounds in sweet basil (Ocimum basilicum L.) leafy vegetable. Scientifica, 2020.
Alabouvette, C., Olivain, C., & Steinberg, C. (2006). Biological control of plant diseases: the European situation. European journal of plant pathology, 114, 329-341.
Alghamdi, B. A., Alshumrani, E. S., Saeed, M. S. B., Rawas, G. M., Alharthi, N. T., Baeshen, M. N., ... & Suhail, M. (2020). Analysis of sugar composition and pesticides using HPLC and GC–MS techniques in honey samples collected from Saudi Arabian markets. Saudi Journal of Biological Sciences, 27(12), 3720-3726.
Ali, A., Shah, L., Rahman, S., Riaz, M. W., Yahya, M., Xu, Y. J., ... & Cheng, B. (2018). Plant defense mechanism and current understanding of salicylic acid and NPRs in activating SAR. Physiological and molecular plant pathology, 104, 15-22.
Alshammari, G. M., Al-Daghri, N. M., & Yakout, S. M. (2021). Difenoconazole and human health: A systematic review of exposure levels, toxicity, and health effects. Journal of Environmental Science and Health, Part C, 39(1), 22-44.
Anderson, R. G., & McDowell, J. M. (2015). A PCR assay for the quantification of growth of the oomycete pathogen Hyaloperonospora arabidopsidis in A rabidopsis thaliana. Molecular Plant Pathology, 16(8), 893-898.
Arabidopsis Genome Initiative. (2000). Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature, 408(6814), 796-815.
Arrus, K., Blank, G., Abramson, D., Clear, R., & Holley, R. A. (2005). Aflatoxin production by Aspergillus flavus in Brazil nuts. Journal of Stored Products Research, 41(5), 513-527.
Atreya, K., Johnsen, F. H., & Sitaula, B. K. (2012). Health and environmental costs of pesticide use in vegetable farming in Nepal. Environment, Development and Sustainability, 14, 477-493.
Ayala, G., Torres, R., & Canseco, M. (2021). Microbial growth curve. In StatPearls [Internet]. StatPearls Publishing.
Bailey, K., Çevik, V., Holton, N., Byrne-Richardson, J., Sohn, K. H., Coates, M., ... & Tör, M. (2011). Molecular cloning of ATR5Emoy2 from Hyaloperonospora arabidopsidis, an avirulence determinant that triggers RPP5-mediated defense in Arabidopsis. Molecular plant-microbe interactions, 24(7), 827-838.
Barrada, A., Delisle-Houde, M., Nguyen, T. T. A., Tweddell, R. J., & Dorais, M. (2022). Drench Application of Soy Protein Hydrolysates Increases Tomato Plant Fitness, Fruit Yield, and Resistance to a Hemibiotrophic Pathogen. Agronomy, 12(8), 1761.
Behind The Bench Staff (2019, September 25). PCR Primer Design Tips. Behind the Bench.
Beketov, M. A., Kefford, B. J., Schäfer, R. B., & Liess, M. (2013). Pesticides reduce regional biodiversity of stream invertebrates. Proceedings of the National Academy of Sciences, 110(27), 11039-11043.
Benami, M., Isack, Y., Grotsky, D., Levy, D., & Kofman, Y. (2020). The economic potential of arbuscular mycorrhizal fungi in agriculture. Grand challenges in fungal biotechnology, 239-279.
Bent, A.F., Innes, R.W., Ecker, J.R. and Staskawicz, B.J. (1992) Disease development in ethylene-insensitive Arabidopsis thaliana infected with virulent and avirulent Pseudomonas and Xanthomonas
Bernoux, M., Cerri, C.E.P., (2005). GEOCHEMISTRY | Soil, Organic Components, Encyclopedia of Analytical Science (Second Edition), Pages 203-208, ISBN 9780123693976,
Bevan, M., Mayer, K., White, O., Eisen, J. A., Preuss, D., Bureau, T., ... & Mewes, H. W. (2001). Sequence and analysis of the Arabidopsis genome. Current opinion in plant biology, 4(2), 105-110.
Bhalerao, R. P., & Fischer, U. (2017). Environmental and hormonal control of cambial stem cell dynamics. Journal of experimental botany, 68(1), 79-87.
Birtić, S., & Kranner, I. (2006). Isolation of high‐quality RNA from polyphenol‐, polysaccharide‐and lipid‐rich seeds. Phytochemical Analysis: An International Journal of Plant Chemical and Biochemical Techniques, 17(3), 144-148.
Bliffeld, M., Mundy, J., Potrykus, I., & Fütterer, J. (1999). Genetic engineering of wheat for increased resistance to powdery mildew disease. Theoretical and Applied Genetics, 98, 1079-1086.
Bocanegra, M. P., Lobartini, J. C., & Orioli, G. A. (2006). Plant uptake of iron chelated by humic acids of different molecular weights. Communications in soil science and plant analysis, 37(1-2), 239-248.
Bock, C. H., Parker, P. E., & Cook, A. Z. (2017). Diseases caused by fungi. In Plant disease epidemiology: Facing challenges of the 21st century (pp. 41-71). Springer.
Bozdogan, A. M. (2014). Assessment of total risk on non-target organisms in fungicide application for agricultural sustainability. Sustainability, 6(2), 1046-1058.
Brouwer, M., Lievens, B., Van Hemelrijck, W., Van den Ackerveken, G., Cammue, B. P., & Thomma, B. P. (2003). Quantification of disease progression of several microbial pathogens on Arabidopsis thaliana using real-time fluorescence PCR. FEMS Microbiology Letters, 228(2), 241-248.
Burns, M. J., Nixon, G. J., Foy, C. A., & Harris, N. (2005). Standardisation of data from real-time quantitative PCR methods–evaluation of outliers and comparison of calibration curves. BMC biotechnology, 5(1), 1-13.
Bustin, S. A. (2010). Why the need for qPCR publication guidelines? --The case for MIQE. Methods, 50(4), 217-226.
Bustin, S. A., Benes, V., Garson, J. A., Hellemans, J., Huggett, J., Kubista, M., ... & Wittwer, C. T. (2009). The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clinical chemistry, 55(4), 611-622.
Bustin, S.A., Benes, V., Garson, J.A., Hellemans, J., Huggett, J., Kubista, M., Mueller, R., Nolan, T., Pfaffl, M.W., Shipley, G.L., et al. (2009). The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 55, 611–622.
Cabral, A., Stassen, J. H., Seidl, M. F., Bautor, J., Parker, J. E., & Van den Ackerveken, G. (2011). Identification of Hyaloperonospora arabidopsidis transcript sequences expressed during infection reveals isolate-specific effectors. PLoS One, 6(5), e19328.
Canellas, L. P., Olivares, F. L., Aguiar, N. O., Jones, D. L., Nebbioso, A., Mazzei, P., & Piccolo, A. (2015). Humic and fulvic acids as biostimulants in horticulture. Scientia Horticulturae, 196, 15–27.
Cao, H., & Shockey, J. M. (2012). Comparison of TaqMan and SYBR Green qPCR methods for quantitative gene expression in tung tree tissues. Journal of agricultural and food chemistry, 60(50), 12296-12303.
Cerdán, M., Sánchez-Sánchez, A., Jordá, D.J., Juárez, M., Andreu, J.S., 2013. Effect of commercial amino acids on iron nutrition of tomato plants grown under lime-induced iron deficiency. J. Plant Nutr. Soil Sci. 176, 1–8.
Chambers, J. E., Greim, H., Kendall, R. J., Segner, H., Sharpe, R. M., & Van Der Kraak, G. (2014). Human and ecological risk assessment of a crop protection chemical: a case study with the azole fungicide epoxiconazole. Critical reviews in toxicology, 44(2), 176-210.
Chatzissavvidis, C., & Therios, I. (2014). Role of algae in agriculture. Seaweeds (Ed. Pomin VH), 1-37.
Chen, J., Feng, L., Ma, W., Jiang, J., Jiang, X., Yang, Y., ... & Liu, X. (2019). Sybr Green-based qPCR for quantitative detection of the membrane transporter gene LmrS in Lactococcus lactis. Journal of microbiological methods, 166, 105734.
Cheng, Z., Wang, J., Hu, X., Zhou, Y., Zhang, W., & Yang, X. (2020). A novel and efficient approach to synthesize 1,2-benzisothiazolin-3-one. Chemical Engineering Journal, 382, 122909.
Chet I and Baker R (1981) Isolation and biocontrol potential of Trichoderma harmatum from soil naturally suppressive to Rhizoctonia solani. Phytopathology 71: 286–290
Chew, A. L., & Maibach, H. I. (1997). 1, 2‐benzisothiazolin‐3‐one (Proxel®): irritant or allergen? A clinical study and literature review. Contact dermatitis, 36(3), 131-136.
Chojnacka, K., Saeid, A., Witkowska, Z., & Tuhy, L. (2012). Biologically active compounds in seaweed extracts-the prospects for the application. In The open conference proceedings journal (Vol. 3, No. 1, pp. 20-28). Bentham Science Publishers Ltd.
Coates, M. E., & Beynon, J. L. (2010). Hyaloperonospora arabidopsidis as a pathogen model. Annual review of phytopathology, 48, 329-345.
Colla, G., Nardi, S., Cardarelli, M., Ertani, A., Lucini, L., Canaguier, R., & Rouphael, Y. (2015). Protein hydrolysates as biostimulants in horticulture. Scientia Horticulturae, 196, 28-38.
Crandall, S. G., Rahman, A., Quesada-Ocampo, L. M., Martin, F. N., Bilodeau, G. J., & Miles, T. D. (2018). Advances in diagnostics of downy mildews: Lessons learned from other oomycetes and future challenges. Plant disease, 102(2), 265-275.
Crowell, D. N., Packard, C. E., Pierson, C. A., Giner, J. L., Downes, B. P., & Chary, S. N. (2003). Identification of an allele of CLA1 associated with variegation in Arabidopsis thaliana. Physiologia Plantarum, 118(1), 29-37.
De Melo, B. A. G., Motta, F. L., & Santana, M. H. A. (2016). Humic acids: Structural properties and multiple functionalities for novel technological developments. Materials Science and Engineering: C, 62, 967-974.
Demmig‐Adams, B., Gilmore, A. M., & Iii, W. W. A. (1996). In vivo functions of carotenoids in higher plants. The FASEB Journal, 10(4), 403-412.
Desjardins, P. R., Zentmyer, G. A., & Reynolds, D. A. (1969). Electron microscopic observations of the flagellar hairs of Phytophthora palmivora zoospores. Canadian Journal of Botany, 47(7), 1077-1079.
Dieffenbach, C. W., & Dveksler, G. S. (2009). PCR primer design. Cold Spring Harbor protocols, 2009(6), pdb-prot5179.
DNeasy PowerSoil Pro Kits. (n.d.). Qiagen.com.
Douglas, P. E., & Flannigan, B. (1988). A microbiological evaluation of barley malt production. Journal of the Institute of Brewing, 94(2), 85-88.
Du Jardin, P. (2015). Plant biostimulants: Definition, concept, main categories and regulation. Scientia horticulturae, 196, 3-14.
Edelmann, H. G., Neinhuis, C., & Bargel, H. (2005). Influence of hydration and temperature on the rheological properties of plant cuticles and their impact on plant organ integrity. Journal of Plant Growth Regulation, 24, 116-126.
El-Ghamry, A. M., Abd El-Hai, K. M., & Ghoneem, K. M. (2009). Amino and humic acids promote growth, yield and disease resistance of faba bean cultivated in clayey soil. Aust. J. Basic Appl. Sci, 3(2), 731-739.
Ertani, A., Cavani, L., Pizzeghello, D., Brandellero, E., Altissimo, A., Ciavatta, C., Nardi, S., 2009. Biostimulant activities of two protein hydrolysates on the growth and nitrogen metabolism in maize seedlings. J. Plant. Nutr. Soil. Sci. 172 – 237.
Ertani, A., Schiavon, M., & Nardi, S. (2017). Transcriptome-wide identification of differentially expressed genes in Solanum lycopersicon L. in response to an alfalfa-protein hydrolysate using microarrays. Frontiers in Plant Science, 8, 1159.
Evans, M. L., & Cleland, R. E. (1985). The action of auxin on plant cell elongation. Critical Reviews in Plant Sciences, 2(4), 317-365.
Fantozzi, E. (2016). Investigating PAMP-triggered immunity in the Arabidopsis thaliana/Hyaloperonospora arabidopsidis interaction (Doctoral dissertation, University of Worcester).
Farrar, J. S., Wittwer, C. T., & Extreme PCR Methods Workshop, M. (2015). Extreme PCR: efficient and specific DNA amplification in the presence of inhibitors. In Extreme PCR (pp. 3-15). Springer.
Fetch Jr, T. G., & Steffenson, B. J. (1999). Rating scales for assessing infection responses of barley infected with Cochliobolus sativus. Plant disease, 83(3), 213-217.
Fiedor, J., & Burda, K. (2014). Potential role of carotenoids as antioxidants in human health and disease. Nutrients, 6(2), 466-488.
Fones, H. N., Bebber, D. P., Chaloner, T. M., Kay, W. T., Steinberg, G., & Gurr, S. J. (2020). Threats to global food security from emerging fungal and oomycete crop pathogens. Nature Food, 1(6), 332-342.
Forrn Mixture, P. Pel| C SH Harrison Road, Airfield Busines5 Park. Signal, 3, 3.
Fuller, S. J., Denyer, S. P., Hugo, W. B., Pemberton, D., Woodcock, P. M., & Buckley, A. J. (1985). The mode of action of l, 2‐benzisothiazolin‐3‐one on Staphylococcus aureus. Letters in applied microbiology, 1(1), 13-15.
Furumura, M. T., Delforno, T. P., Cabral, E., & de Oliveira, L. M. (2018). Sublethal concentrations of biocides increase antibiotic resistance in swine Escherichia coli isolates. Microbial Pathogenesis, 114, 291-296.
Gadjev, I., Vanderauwera, S., Gechev, T. S., Laloi, C., Minkov, I. N., Shulaev, V., ... & Van Breusegem, F. (2006). Transcriptomic footprints disclose specificity of reactive oxygen species signaling in Arabidopsis. Plant physiology, 141(2), 436-445.
García-Angulo, V. A. (2017). Overcoming the barriers of plant immunity by Salmonella. Virulence, 8(7), 1350-1354.
García-Sánchez, F., García-Sánchez, R., Mounzer, O., & Noguera, V. (2018). Effect of autoclaving on the activity of a commercial biostimulant based on seaweed extracts. Journal of Plant Nutrition and Soil Science, 181(1), 147-154.
Gessler, C., Pertot, I., and Perazzolli, M. 2011.Plasmopara viticola: a review ofknowledge on downy mildew of grapevine and effective disease management. Phytopathol. Mediterr. 50:3-44.
Gisi, U. (2002). Chemical control of downy mildews. Advances in downy mildew research, 119-159.
GMO legislation. (n.d.). Food Safety.
González-Rodríguez, R. M., Rial-Otero, R., Cancho-Grande, B., & Simal-Gándara, J. (2008). Occurrence of fungicide and insecticide residues in trade samples of leafy vegetables. Food Chemistry, 107(3), 1342-1347.
Graham, J. H., & Madden, L. V. (2014). Interpretation of disease severity values. Phytopathology, 104(1), 3-6.
Gruvberger, B., Isaksson, M., & Julander, A. (2018). Allergic contact dermatitis caused by 1,2-benzisothiazolin-3-one in a washing machine cleaner. Contact Dermatitis, 78(4), 262-263.
Guillemaut C (2003) Identification et e´tude de l’e´cologie de Rhizoctonia solani, responsable de la maladie de pourriture brune de la betterave sucrie`re. PhD Thesis Universite´ Claude Bernard Lyon. 90 p.
Gupt, S. K., Chand, R., Mishra, V. K., Ahirwar, R. N., Bhatta, M., & Joshi, A. K. (2021). Spot blotch disease of wheat as influenced by foliar trichome and stomata density. Journal of Agriculture and Food research, 6, 100227.
Hammerschmidt, R. (2009). Systemic acquired resistance. Advances in botanical research, 51, 173-222.
Hardy, G., & McComb (2008), J. Project Period and Period Covered by this Report.
Havaux, M., & Kloppstech, K. (2001). The protective functions of carotenoid and flavonoid pigments against excess visible radiation at chilling temperature investigated in Arabidopsis npq and tt mutants. Planta, 213, 953-966.
Hoffman, T., Schmidt, J.S., Zheng, X. and Bent, A.F. (1999) Isolation of ethylene-insensitive soybean mutants that are altered in pathogen susceptibility and gene-for-gene disease resistance. Plant Physiol. 119, 935-950
Horsfall, J. G., & Barratt, R. W. (1945). An improved grading system for measuring plant diseases. Phytopathology, 35(8), 655-665.
Hua, M., Liu, X., Ma, Y., Zhang, Z., Wang, Z., Sun, X., ... & Wu, J. (2020). Distribution and fate of difenoconazole in greenhouse vegetable soil-water system. Environmental Pollution, 258, 113722.
Huibers, R.P. , de Jong, M. , Dekter, R.W. and Van den Ackerveken, G. (2009) Disease‐specific expression of host genes during downy mildew infection of Arabidopsis. Mol. Plant–Microbe Interact. 22, 1104–1115.
IBM Corp. (2020). IBM SPSS Statistics for Windows (Version 27.0). Armonk, NY: IBM Corp.
Ioio, R. D., Linhares, F. S., Scacchi, E., Casamitjana-Martinez, E., Heidstra, R., Costantino, P., & Sabatini, S. (2007). Cytokinins determine Arabidopsis root-meristem size by controlling cell differentiation. Current biology, 17(8), 678-682.
Isenring, R. (2010). Pesticides and the loss of biodiversity. Pesticide Action Network Europe, London, 26.
iTaq Universal SYBR Green SuperMix. (n.d.). Bio-Rad Laboratories.
Kamran, S., Sinniah, A., Abdulghani, M. A., & Alshawsh, M. A. (2022). Therapeutic potential of certain terpenoids as anticancer agents: a scoping review. Cancers, 14(5), 1100.
Kaur, M., Bhari, R., & Singh, R. S. (2021). Chicken feather waste-derived protein hydrolysate as a potential biostimulant for cultivation of mung beans. Biologia, 76, 1807-1815.
Kim, H. J., Yoo, D. H., Kim, D. H., & Lee, C. H. (2014). Interaction of 1,2-benzisothiazolin-3-one with metal ions and its effect on metalloenzymes. Journal of Enzyme Inhibition and Medicinal Chemistry, 29(5), 617-623.
Kim, T. K. (2015). T test as a parametric statistic. Korean journal of anesthesiology, 68(6), 540-546.
Knapp, J. S., & Bromley-Challoner, K. C. A. (2003). Recalcitrant organic compounds. Handbook of water and wastewater microbiology, 559-595.
Koch, E., & Slusarenko, A. (1990). Arabidopsis is susceptible to infection by a downy mildew fungus. The Plant Cell, 2(5), 437-445.
Koornneef, M., & Meinke, D. (2010). The development of Arabidopsis as a model plant. The Plant Journal, 61(6), 909-921.
Kudela, V. (2009). Potential impact of climate change on geographic distribution of plant pathogenic bacteria in Central Europe. Plant Protection Science, 45(Special Issue).
Küpers, J. J., Oskam, L., & Pierik, R. (2020). Photoreceptors regulate plant developmental plasticity through auxin. Plants, 9(8), 940.
Lachhab, N., Sanzani, S. M., Bahouaoui, M. A., Boselli, M., & Ippolito, A. (2016). Effect of some protein hydrolysates against gray mould of table and wine grapes. European Journal of Plant Pathology, 144, 821-830.
Lahlali, R., Ezrari, S., Radouane, N., Kenfaoui, J., Esmaeel, Q., El Hamss, H., ... & Barka, E. A. (2022). Biological control of plant pathogens: A global perspective. Microorganisms, 10(3), 596.
Lamberth, C., Rendine, S., & Sulzer-Mosse, S. (2021). Agrochemical disease control: The story so far. In Recent Highlights in the Discovery and Optimization of Crop Protection Products (pp. 65-85). Academic Press
Lapertot, M., Pulgarín, C., Fernández-Ibáñez, P., Maldonado, M. I., Pérez-Estrada, L., Oller, I., ... & Malato, S. (2006). Enhancing biodegradability of priority substances (pesticides) by solar photo-Fenton. Water research, 40(5), 1086-1094.
Lebeda, A., & Cohen, Y. (2011). Cucurbit downy mildew (Pseudoperonospora cubensis)—biology, ecology, epidemiology, host-pathogen interaction and control. European journal of plant pathology, 129, 157-192.
Leboldus, J. M., & Isabel, N. (2018). Quantitative polymerase chain reaction (qPCR) detection and quantification of foliar fungal pathogens of poplar. In Poplar and Willow Culture (pp. 221-234). Springer.
Lee, M. H., Jeon, H. S., Kim, S. H., Chung, J. H., Roppolo, D., Lee, H. J., ... & Park, O. K. (2019). Lignin‐based barrier restricts pathogens to the infection site and confers resistance in plants. The EMBO journal, 38(23), e101948.
Lhermie, G., Sauvage, P., Tauer, L. W., Chiu, L. V., Kanyiamattam, K., Ferchiou, A., ... & Grohn, Y. T. (2020). Economic effects of policy options restricting antimicrobial use for high risk cattle placed in US feedlots. PLoS One, 15(9), e0239135.
Lin, Y., Wang, X., Zhang, X., Song, Y., & Xie, X. (2021). Prenatal exposure to difenoconazole disturbs placental amino acid transport and causes fetal growth retardation in mice. Environmental Pollution, 273, 116450.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. methods, 25(4), 402-408.
Lu, L., Guo, Y., & Wang, X. (2021). Understanding bacterial growth curve and applications in microbiology. Frontiers in Microbiology, 12, 652069.
Lucini, L., Miras-Moreno, B., Rouphael, Y., Cardarelli, M., & Colla, G. (2020). Combining molecular weight fractionation and metabolomics to elucidate the bioactivity of vegetal protein hydrolysates in tomato plants. Frontiers in Plant Science, 11, 976.
Madden, L. V., Hughes, G., & van den Bosch, F. (2007). The study of plant disease epidemics. American Phytopathological Society.Magyarosy,
Mahmood, I., Imadi, S. R., Shazadi, K., Gul, A., & Hakeem, K. R. (2016). Effects of pesticides on environment. Plant, soil and microbes: volume 1: implications in crop science, 253-269.
Mandal, K., Saravanan, R., Maiti, S., & Kothari, I. L. (2009). Effect of downy mildew disease on photosynthesis and chlorophyll fluorescence in Plantago ovata Forsk. Journal of Plant Diseases and Protection, 116(4), 164.
Mattioli, R., Francioso, A., d’Erme, M., Trovato, M., Mancini, P., Piacentini, L., ... & Mosca, L. (2019). Anti-inflammatory activity of a polyphenolic extract from Arabidopsis thaliana in in vitro and in vivo models of Alzheimer’s disease. International journal of molecular sciences, 20(3), 708.
McDowell, J. M., Hoff, T., Anderson, R. G., & Deegan, D. (2011). Propagation, storage, and assays with Hyaloperonospora arabidopsidis: a model oomycete pathogen of Arabidopsis. Plant Immunity: Methods and Protocols, 137-151.
Meinke, D. W., Cherry, J. M., Dean, C., Rounsley, S. D., & Koornneef, M. (1998). Arabidopsis thaliana: a model plant for genome analysis. Science, 282(5389), 662-682.
Meléndrez, M. M. (2020). Humic acid: The science of humus and how it benefits soil. Eco Farming Daily
Microvolume spectrofotometer, mySPEC. (n.d.). VWR.
Miedes, E., Vanholme, R., Boerjan, W., & Molina, A. (2014). The role of the secondary cell wall in plant resistance to pathogens. Frontiers in plant science, 5, 358.
Mok, M. C. (2019). Cytokinins and plant development—an overview. Cytokinins, 155-166.
Mondino, P., Casanova, L., Celio, A., Bentancur, O., Leoni, C., & Alaniz, S. (2015).
Sensitivity of Venturia inaequalis to trifloxystrobin and difenoconazole in Uruguay. Journal of Phytopathology, 163(1), 1-10.
Mrid, R. B., Benmrid, B., Hafsa, J., Boukcim, H., Sobeh, M., & Yasri, A. (2021). Secondary metabolites as biostimulant and bioprotectant agents: A review. Science of The Total Environment, 777, 146204.
Mu, X., Pang, S., Sun, X., Gao, J., Chen, J., Chen, X., ... & Wang, C. (2013). Evaluation of acute and developmental effects of difenoconazole via multiple stage zebrafish assays. Environmental Pollution, 175, 147-157.
Mészáros, É. (2022, May 11). qPCR: How SYBR® Green and TaqMan® real-time PCR assays work. INTEGRA.
Niemeier, R. T., Sivasubramani, S. K., Reponen, T., & Grinshpun, S. A. (2006). Assessment of fungal contamination in moldy homes: comparison of different methods. Journal of occupational and environmental hygiene, 3(5), 262-273.
NISHI, A., & KUROSAKI, F. (2012). of Carotenoids and Phytoalexins. Medicinal and Aromatic Plants V, 24, 178.
Oshchepkov, M. S., Kalistratova, A. V., Savelieva, E. M., Romanov, G. A., Bystrova, N. A., & Kochetkov, K. A. (2020). Natural and synthetic cytokinins and their applications in biotechnology, agrochemistry and medicine. Russian Chemical Reviews, 89(8), 787.
Palta, J. P. (1990). Leaf chlorophyll content. Remote sensing reviews, 5(1), 207-213.
Palumbo, G., Schiavon, M., Nardi, S., Ertani, A., Celano, G., & Colombo, C. M. (2018). Biostimulant potential of humic acids extracted from an amendment obtained via combination of olive mill wastewaters (OMW) and a pre-treated organic material derived from municipal solid waste (MSW). Frontiers in Plant Science, 9, 1028.
Pandey, A. K., Pandey, S. D., & Misra, V. (2000). Stability constants of metal–humic acid complexes and its role in environmental detoxification. Ecotoxicology and environmental safety, 47(2), 195-200.
Patil, B., & Chetan, H. T. (2018). Foliar fertilization of nutrients. Marumegh, 3(1), 49-53.
Paul, K., Sorrentino, M., Lucini, L., Rouphael, Y., Cardarelli, M., Bonini, P., Reynaud, H., Canaguier, R., Trtílek, M., Panzarová, K., & Colla, G. (2019). Understanding the Biostimulant Action of Vegetal-Derived Protein Hydrolysates by High-Throughput Plant Phenotyping and Metabolomics: A Case Study on Tomato. Frontiers in Plant Science, 10.
Pel, M. J., Wintermans, P. C., Cabral, A., Robroek, B. J., Seidl, M. F., Bautor, J., ... & Pieterse, C. M. (2014). Functional analysis of Hyaloperonospora arabidopsidis RXLR effectors. PloS one, 9(11), e110624.
Pfeufer, E., & Harrison, L. (2022). High incidence of Hyaloperonospora cardamines‐laciniatae on Cardamine concatenata in Maryland, USA. New Disease Reports, 46(2).
Pimentel, D., Acquay, H., Biltonen, M., Rice, P., Silva, M., Nelson, J., ... & D'amore, M. (1992). Environmental and economic costs of pesticide use. BioScience, 42(10), 750-760.
Piubelli, G. C., Hoffmann-Campo, C. B., Moscardi, F., Miyakubo, S. H., & Neves De Oliveira, M. C. (2005). Are chemical compounds important for soybean resistance to Anticarsia gemmatalis?. Journal of Chemical ecology, 31, 1509-1525.
Prilutskaya, N., Popova, L., Korelskaya, T., Nikitina, M., & Gerasimova, L. (2019). The estimation of the humus acids structural and composition in the soils of the euro-arctic zone and their ecoprotective role to trace metals. International Multidisciplinary Scientific GeoConference: SGEM, 19(3.2), 467-474.
Prince, A., Roy, S., & McDonald, D. (2022). Exploration of the antimicrobial synergy between selected natural substances on Streptococcus mutans to identify candidates for the control of dental caries. Microbiology Spectrum, 10(3), e02357-21.
Quan, H., Li, H., Zhang, X., Huang, K., & Li, P. (2021). Growth curve and cell cycle analysis of Streptococcus suis. BMC Microbiology, 21(1), 44.
R Core Team (2013). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0
Rausher, M. D. (2001). Co-evolution and plant resistance to natural enemies. Nature, 411(6839), 857-864.
Ried, M. K., Banhara, A., Hwu, F. Y., Binder, A., Gust, A. A., Höfle, C., ... & Parniske, M. (2019). A set of Arabidopsis genes involved in the accommodation of the downy mildew pathogen Hyaloperonospora arabidopsidis. PLoS pathogens, 15(7), e1007747.
Roberts, C. A. (1995). Microbiology of stored forages. Post‐harvest physiology and preservation of forages, 22, 21-38.
Rossi, V., Caffi, T., Giosuè, S., & Bugiani, R. (2008). A mechanistic model simulating primary infections of downy mildew in grapevine. Ecological modelling, 212(3-4), 480-491.
Rouabhi, R. (2010). Introduction and toxicology of fungicides. INTECH Open Access Publisher.
Rouphael, Y., Cardarelli, M., Bonini, P., & Colla, G. (2017). Synergistic action of a microbial-based biostimulant and a plant- derived protein hydrolysate enhances lettuce tolerance to alkalinity and salinity. Frontiers in Plant Science, 8, 131.
Rychlik, W. (2007). Selection of primers for polymerase chain reaction. In PCR primer design (pp. 31-40). Humana Press.
Santos, M. S., Nogueira, M. A., & Hungria, M. (2019). Microbial inoculants: reviewing the past, discussing the present and previewing an outstanding future for the use of beneficial bacteria in agriculture. Amb Express, 9, 1-22.
Saunders, H., & Watkins, F. (2001). Allergic contact dermatitis due to thiuram exposure from a fungicide. Australasian journal of dermatology, 42(3), 217-218.
Savary, S., Willocquet, L., Elazegui, F. A., Castilla, N. P., & Teng, P. S. (2012). Rice pest constraints in tropical Asia: quantification of yield losses due to rice pests in a range of production situations. Plant Disease, 96(5), 596-604.
Schena, L., Nigro, F., & Ippolito, A. (2013). Rapid detection methods for plant pathogens: principles, applications, and prospects. In Rapid Detection of Infectious Agents (pp. 267-295). Springer.
Schikora, A., Carreri, A., Charpentier, E., & Hirt, H. (2008). The dark side of the salad: Salmonella typhimurium overcomes the innate immune response of Arabidopsis thaliana and shows an endopathogenic lifestyle. PLoS One, 3(5), e2279.
Schoina, C., & Govers, F. (2015). The oomycete Phytophthora infestans, the irish potato famine pathogen. Principles of Plant-Microbe Interactions: Microbes for Sustainable Agriculture, 371-378.
Shimizu-Sato, S., Tanaka, M., & Mori, H. (2009). Auxin–cytokinin interactions in the control of shoot branching. Plant molecular biology, 69, 429-435.
Shukla, P. S., Borza, T., Critchley, A. T., & Prithiviraj, B. (2021). Seaweed-based compounds and products for sustainable protection against plant pathogens. Marine drugs, 19(2), 59.
Sible, C. N., Seebauer, J. R., & Below, F. E. (2021). Plant biostimulants: A categorical review, their implications for row crop production, and relation to soil health indicators. Agronomy, 11(7), 1297.
Singer, V. L., Lawlor, T. E., & Yue, S. (1999). Comparison of SYBR® Green I nucleic acid gel stain mutagenicity and ethidium bromide mutagenicity in the Salmonella/mammalian microsome reverse mutation assay (Ames test). Mutation Research/Genetic Toxicology and Environmental Mutagenesis, 439(1), 37-47.
Smilkova, M., Smilek, J., Kalina, M., Klucakova, M., Pekar, M., & Sedlacek, P. (2019). A simple technique for assessing the cuticular diffusion of humic acid biostimulants. Plant methods, 15, 1-11.
Steelink, C. (1963). What is humic acid?.
Stevenson, F.J., 1994. Humus Chemistry: Genesis, Composition, Reaction (Second ed.), Wiley, New York.
Stirk, W. A., & Van Staden, J. (1996). Comparison of cytokinin-and auxin-like activity in some commercially used seaweed extracts. Journal of Applied Phycology, 8, 503-508.
Stoytcheva, M. (Ed.). (2011). Pesticides in the modern world: trends in pesticides analysis. BoD–Books on Demand.
Telli, O., Jimenez‐Quiros, C., McDowell, J. M., & Tör, M. (2020). Effect of light and dark on the growth and development of downy mildew pathogen Hyaloperonospora arabidopsidis. Plant Pathology, 69(7), 1291-1300.
Thakur, R. P., & Mathur, K. (2002). Downy mildews of India. Crop Protection, 21(4), 333-345.
Thines, M. (2018). Oomycetes. Current Biology, 28(15), R812-R813.
Tomé, D. F., Steinbrenner, J., & Beynon, J. L. (2014). A growth quantification assay for Hyaloperonospora arabidopsidis isolates in Arabidopsis thaliana. Plant-Pathogen Interactions: Methods and Protocols, 145-158.
Travadon, R., Rolshausen, P. E., Gubler, W. D., Cadle-Davidson, L., & Baumgartner, K. (2013). Susceptibility of cultivated and wild Vitis spp. to wood infection by fungal trunk pathogens. Plant Disease, 97(12), 1529-1536.
Tremblay, A., Seabolt, S., Zeng, H., Zhang, C., Böckler, S., Tate, D. N., ... & Lu, H. (2016). A role of the Fuzzy Onions Like gene in regulating cell death and defense in Arabidopsis. Scientific Reports, 6(1), 37797.
Tsuchiya, H., Ohta, T., & Masuda, S. (2015). Optimization of qPCR primer design using in silico PCR software and its application for detecting SNPs in a gene involved in iron metabolism. BMC research notes, 8(1), 1-7.
USDA. 2015. United States Department of Agriculture, National Agricultural Statistics Service, Crop Production 2014 Summary, Vegetables 2014 Summary, and Quick Stats.
Van Den Bosch, F., Paveley, N., Shaw, M., Hobbelen, P., & Oliver, R. (2011). The dose rate debate: does the risk of fungicide resistance increase or decrease with dose?. Plant Pathology, 60(4), 597-606.
Vega, K., & Kalkum, M. (2012). Chitin, chitinase responses, and invasive fungal infections. International journal of microbiology, 2012.
Venbrux, M., Crauwels, S., & Rediers, H. (2023). Current and emerging trends in techniques for plant pathogen detection. Frontiers in Plant Science, 8, 1186.
Vernieri, P., Borghesi, E., Ferrante, A., & Magnani, G. (2005). Application of biostimulants in floating system for improving rocket quality. Journal of Food Agriculture and Environment, 3(3/4), 86.
Voiculescu, D. I., Roman, D. L., Ostafe, V., & Isvoran, A. (2022). A cheminformatics study regarding the human health risks assessment of the stereoisomers of difenoconazole. Molecules, 27(15), 4682.
Walters, D. R., & McRoberts, N. (2006). Plants and biotrophs: a pivotal role for cytokinins?. Trends in plant science, 11(12), 581-586.
Wang, R. K., Wang, C. E., Fei, Y. Y., Gai, J. Y., & Zhao, T. J. (2013). Genome-wide identification and transcription analysis of soybean carotenoid oxygenase genes during abiotic stress treatments. Molecular biology reports, 40, 4737-4745.
Wang, Y., Li, X., Fan, B., Zhu, C., & Chen, Z. (2021). Regulation and function of defense-related callose deposition in plants. International Journal of Molecular Sciences, 22(5), 2393.
Wang, Y., Liu, Z., Zhang, Y., & Guo, H. (2019). Effects of 1,2-benzisothiazolin-3-one on glutathione peroxidase activity and oxidative stress in human neuroblastoma cells. Environmental Toxicology and Pharmacology, 67, 60-65.
Wang, Y., Pruitt, R. N., Nürnberger, T., & Wang, Y. (2022). Evasion of plant immunity by microbial pathogens. Nature Reviews Microbiology, 20(8), 449-464.
Wang, Z., Hong, X., Hu, K., Wang, Y., Wang, X., Du, S., ... & Li, Y. (2017). Impaired magnesium protoporphyrin IX methyltransferase (ChlM) impedes chlorophyll synthesis and plant growth in rice. Frontiers in plant science, 8, 1694.
Weller DM and Thomashow LS (1993) Microbial metabolites with biological activity. In: Lumsden D and Vaugh JL (eds.) Pest Management: Biologically Based Technologies (pp. 173–180) American Chemical Society, Washington, USA
Whisson, S. C., Boevink, P. C., Moleleki, L., Avrova, A. O., Morales, J. G., Gilroy, E. M., ... & Birch, P. R. (2007). A translocation signal for delivery of oomycete effector proteins into host plant cells. Nature, 450(7166), 115-118.
Worthington, P. (2012). Sterol biosynthesis inhibiting triazole fungicides. Bioactive heterocyclic compound classes, 129.
Ye, J., Coulouris, G., Zaretskaya, I., Cutcutache, I., Rozen, S., & Madden, T. L. (2012). Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC bioinformatics, 13(1), 1-11.
Zacchino, S. A., Butassi, E., Di Liberto, M., Raimondi, M., Postigo, A., & Sortino, M. (2017). Plant phenolics and terpenoids as adjuvants of antibacterial and antifungal drugs. Phytomedicine, 37, 27-48.
Zhang, Y. J., Xie, Z. K., Wang, Y. J., Su, P. X., An, L. P., & Gao, H. (2011). Effect of water stress on leaf photosynthesis, chlorophyll content, and growth of oriental lily. Russian Journal of Plant Physiology, 58, 844-850.
Zodape, S. T., Gupta, A., Bhandari, S. C., Rawat, U. S., Chaudhary, D. R., Eswaran, K., & Chikara, J. (2011). Foliar application of seaweed sap as biostimulant for enhancement of yieldand quality of tomato (Lycopersicon esculentum Mill.).









